Pharmaceutical manufacturing is entering a new phase of digital acceleration. Across the industry, we observe a clear pattern: Organizations that have historically lagged in factory modernization are now showing stronger interest in Digital Factory Transformation initiatives. This shift is encouraging. Improving factory visibility is a critical first step toward addressing long-standing productivity challenges and building a foundation for operational excellence. However, as momentum grows, it becomes equally important to ensure that this transformation is done right the first time.
One of the most significant barriers to successful digital transformation is the lack of harmonization in manufacturing KPI tracking—particularly in Overall Equipment Effectiveness (OEE)—as well as inconsistencies in digital performance systems. As pharma manufacturers scale digital initiatives across multiple sites and production lines, the need for standardized, reliable, and granular performance data becomes essential. Without harmonization, even the most advanced digital solutions fail to deliver meaningful, actionable insights. This negatively impacts ROI.
In this article, we explore the critical role of harmonizing OEE and digital performance in enabling effective digital transformation. We examine how this foundation supports operational excellence, aligns with world-class manufacturing principles, and enables advanced capabilities such as AI-driven manufacturing. We also highlight practical examples from our Digital Factory Platform to illustrate what is achievable when harmonization is implemented effectively.
The Hidden Complexity of OEE in Pharma: Beyond the Surface Metric
While OEE is globally recognized as the gold standard for measuring manufacturing productivity, its application in the pharmaceutical sector is fraught with inconsistency. In practice, OEE is far from standardized. Different organizations and frequently different departments within the same facility often utilize divergent calculation methodologies, leading to a fragmented view of reality.

The Divergence: OEE1, OEE2, and OEE3
The discrepancy usually begins with how “losses” are defined and categorized. Organizations typically navigate between three primary lenses:
- OEE1: Focused on purely operational time, often excluding specific planned downtimes.
- OEE2: A broader view that includes more loss categories to capture a wider range of inefficiencies.
- OEE3: The most comprehensive “Total Equipment Effectiveness” view, incorporating all losses, including every planned activity.
At first glance, these variations might seem like minor accounting preferences. In reality, they create massive structural discrepancies in how performance is interpreted.
The "Apples to Oranges" Dilemma
Consider a scenario where one production line reports 40% OEE while another reports 60%. Without harmonization, the 40% line may actually be the superior operation. The difference often hinges on how high-frequency pharma activities are recorded. Critical processes such as line cleaning, complex equipment setup, and rigorous logbook/batch reporting may be excluded from the denominator in one site but counted as a major loss in another.
Without a harmonized standard:
- Historical Data Loses Credibility: Long-term trends become impossible to track if the “yardstick” changes over time.
- Cross-Site Benchmarking Becomes Meaningless: Global leaders cannot identify their true “top performers” or “laggards.”
- Root Cause Analysis Produces Misleading Conclusions: Teams may end up solving the wrong problems based on skewed data.
- AI Readiness is Compromised: Machine learning models cannot be trained effectively on inconsistent datasets; they require a “single version of the truth.”

The SCW.AI Solution: OEE as a Strategic Compass
For pharma manufacturers aiming to scale Industry 4.0 initiatives, harmonization is a prerequisite. Our Digital Factory Platform addresses this complexity by allowing manufacturers to define any loss item specific to their unique operations while maintaining global consistency.
Our tools empower producers to customize the definitions of OEE1, OEE2, and OEE3 in a strictly harmonized manner across the entire enterprise. This ensures that OEE data functions as a reliable compass for the organization. With this foundation, manufacturers can:
- Conduct precise root cause analysis based on objective data.
- Execute line-by-line or site-by-site comparisons with total confidence.
- Instantly identify high-performing timelines to replicate best practices.
- Pinpoint underperforming assets at a glimpse to prioritize interventions.

Download Our Expert Resource: To master these complexities, download our free guide: The Pharma Manufacturer’s Complete OEE Handbook. Learn the nuances of pharmaceutical OEE calculation, benchmark your performance against industry peers, and discover practical, site-specific strategies for driving measurable OEE improvements.
Beyond Metrics: Harmonizing Actions Across the Organization
World-class manufacturing systems are built on consistent working methods and shared best practices. Digital tools now make it possible to scale this standardization faster and more effectively than ever before.
With digital performance solutions, organizations can:
- Deploy digital work instructions across sites instantly
- Implement Digital Kamishibai systems to audit and sustain standards
- Share proven operational strategies between high-performing and underperforming lines
This creates a powerful feedback loop:
- Harmonized data identifies what works
- Standardized actions replicate success
- Continuous improvement becomes scalable
The result is not just better performance; but consistent performance everywhere.
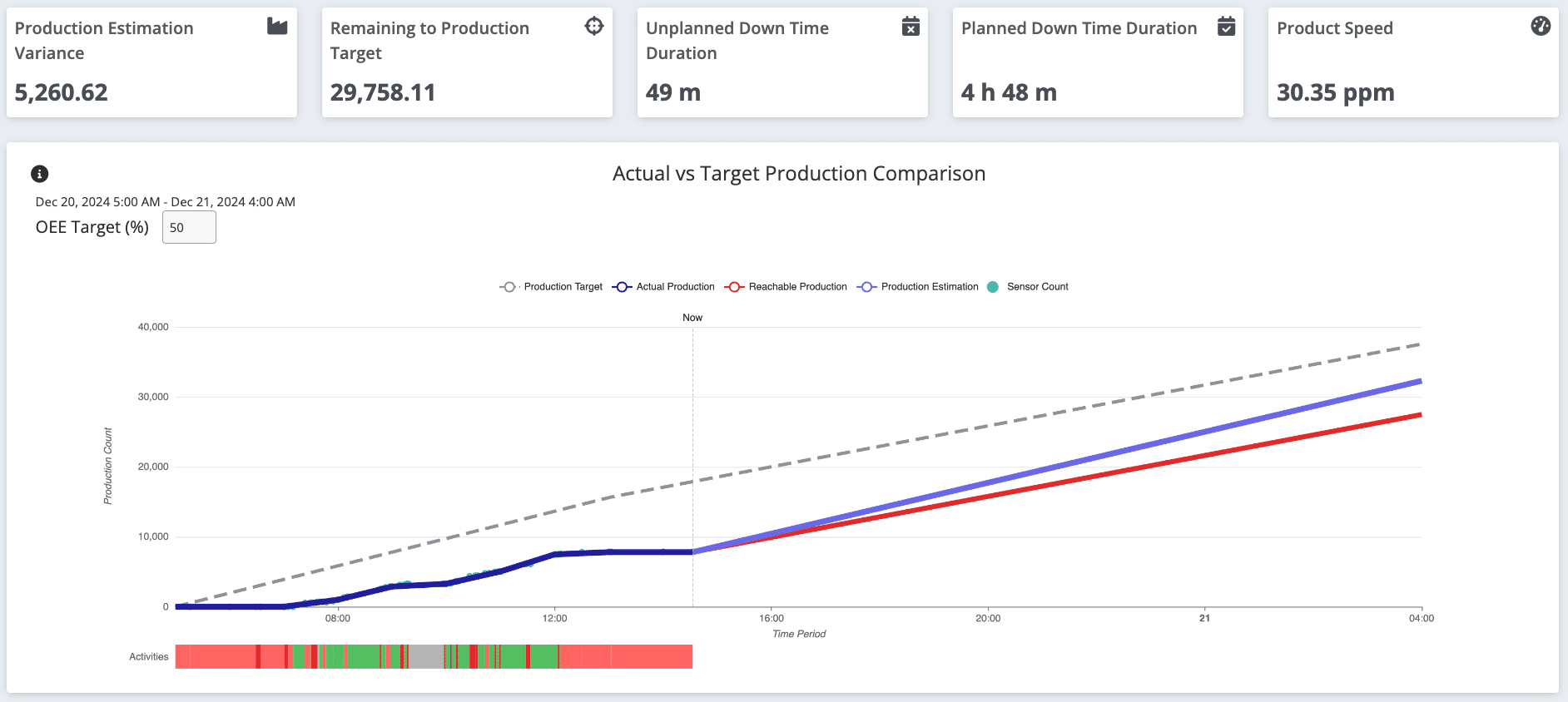
The Critical Role of Granularity
While harmonization ensures consistency, granularity ensures usability.
Pharma manufacturing environments are inherently complex, with multiple variables affecting performance at any given time. High-level KPIs alone are not enough to drive meaningful improvements.
Granular visibility enables organizations to:
- Gain a 360° view of operations using standardized metrics
- Drill down into specific machines, lines, or shifts
- Identify micro-level inefficiencies that aggregate into major losses
- Detect hidden bottlenecks that are otherwise invisible
For example, instead of simply knowing that a site is underperforming, teams can pinpoint:
- Which line is causing the issue
- During which shift the losses occur
- Which work station or process step is responsible
This level of insight transforms data from a reporting tool into a decision-making engine.
The Convergence of Harmonization and Granularity: Enabling True Scientific Management
When global harmonization is paired with granular data, pharmaceutical manufacturers unlock a new echelon of operational transparency. This combination moves the organization beyond mere “tracking” and into the realm of Scientific Management, where decisions are dictated by precise data rather than intuition or outdated reports.
Organizations can:
- Compare factories side-by-side using consistent KPIs
- Establish global benchmarks for productivity and quality
- Track work station to global level performance in real time
- Identify best-performing sites, departments or lines and replicate their success with Digital Work Instructions and execution ready digital knowledgebase
This capability is especially valuable for multinational pharma companies managing complex production networks. Instead of relying on delayed or inconsistent reports, decision-makers gain immediate and reliable visibility into performance across the entire organization.

Our analysis shows that implementing harmonized digitalization and unleashing scientific management can increase OEE drastically. For a typical pharma manufacturer, OEE usually sits between 35% and 40%. However, for harmonized digital factories, this can rise to 55% or 65% within months. Such an improvement can unlock over 20% throughput growth and cost savings simultaneously, providing a crucial competitive advantage.
UP TO
INCREASE IN
OEE
UP TO
INCREASE IN
THROUGHPUT
UP TO
DECREASE IN
PRODUCTION COST
Making Daily Operations Easier for Every Role
One of the most important benefits of harmonized digital performance solutions is their impact on daily operations. When implemented correctly, these systems simplify workflows and empower every role within the factory.

Operators
Operators are often burdened with manual reporting and the confusion of determining which tasks to prioritize and how to execute them. This is an ineffective use of their time and is highly prone to error. With harmonized digital transformation:
- Manual input is minimized or eliminated.
- Data accuracy improves significantly.
- Operators can easily see which tasks are assigned to them, check deadlines, and access clear work instructions.
This not only improves efficiency but also reduces frustration on the shop floor.

Supervisors
Supervisors must react quickly to production issues, yet they often lack real-time visibility and effective execution tools. Harmonized digital systems provide:
- Instant alerts when performance deviates from the plan.
- Line-specific KPIs that highlight problems before they escalate.
- Faster root cause identification and quicker task assignment.
- Accountable tracking of every process.
This enables supervisors to resolve issues proactively rather than reactively.
Mid-Level Managers
At the department or plant level, decision-makers require a view of operations that is both consolidated and detailed. With harmonized and granular data:
- Performance across lines and departments can be monitored in real time.
- Tier meetings become more data-driven and effective using Digital SQCDP Boards.
- Continuous improvement initiatives are based on facts, not assumptions.
This supports a more scientific approach to operational management.
Top Managers
For multi-site operations, the challenge shifts to coordination and resource allocation.
Harmonized data allows top managers to:
- Compare sites objectively
- Identify systemic bottlenecks
- Allocate resources more effectively
- Scale successful strategies across the network
This accelerates standardization and drives global performance improvements.

Executives
At the executive level, the focus is on long-term value and strategic decision-making.
With reliable, harmonized data:
- ROI from digital investments becomes measurable
- Long-term performance trends are clearly visible
- Capital planning decisions are supported by data
This reduces risk and increases confidence in strategic initiatives.
Preparing for AI-Driven Manufacturing
Artificial intelligence is rapidly becoming a cornerstone of advanced manufacturing. However, its success depends entirely on the quality of underlying data.
Harmonized OEE and performance metrics provide:
- Clean, structured datasets for model training
- Consistent definitions of losses and performance
- Reliable historical trends
Without this foundation, AI initiatives are likely to fail or deliver misleading results. In this sense, harmonization is not just about today’s performance—it is about future-proofing the organization.

To learn more about AI applications in pharmaceutical manufacturing, including the potential return on investment and how to prepare your factory for reliable AI deployment, you can download our AI in Pharma White Paper.

Take the First Step Toward a Harmonized Digital Future
Do not let fragmented data hold your productivity back. Build your factory of the future on a foundation of truth.
Achieving a “Right First Time” digital transformation requires more than just new software. At SCW.AI, we help the world’s leading pharma manufacturers move past the inconsistent OEE, providing the granular visibility, harmonized metrics and connected monitoring, execution and planning solutions needed to unlock true operational excellence.
Ready to see how harmonization can transform your global operations? Speak with our industry specialists about your specific OEE challenges and your roadmap to Pharma 4.0.
Book a Demo Today and see our Digital Factory Platform in action and discover how we standardize performance across complex production networks.


